 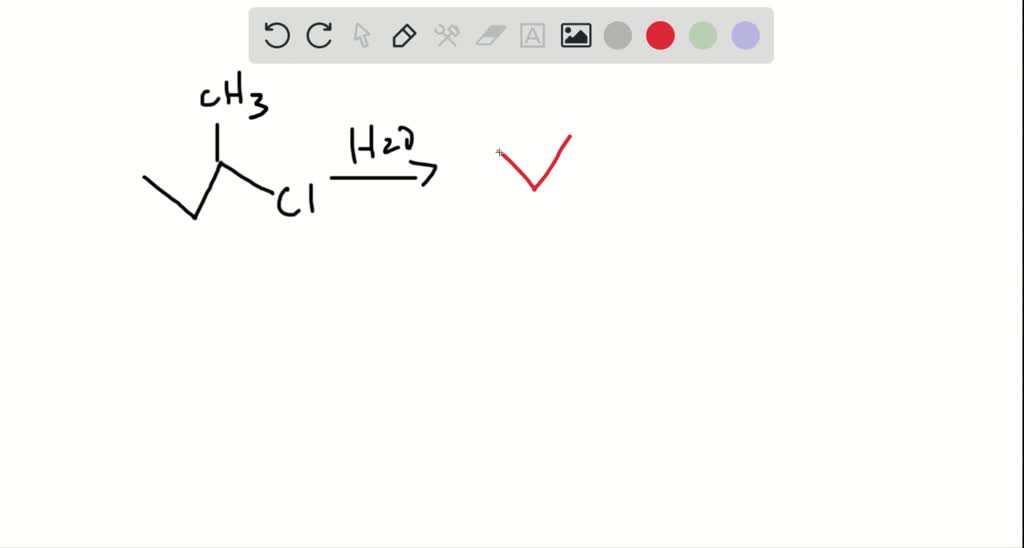
The pK a of a simple alcohol (e.g., MeOH = 15.5, EtOH = 15.9) are close to the value for water (15. These O atoms can react with O 2 molecules to form ozone, O 3, O 2 ( g) + O ( g) O 3 ( g) whose Lewis structure is shown in the figure below. In chemistry, a hydroxy or hydroxyl group is a functional group with the chemical formula OH and composed of one oxygen atom covalently bonded to one hydrogen atom. Hydroxide ions are polyatomic ions with a negative charge, consisting of hydrogen and oxygen. In the presence of lightning or another source of a spark, O 2 molecules dissociate to form oxygen atoms. Thus the pH at which - is formed relative to - will depend on the relative acidity of the alcohol. The Chemistry of Ozone The O 2 molecule is not the only elemental form of oxygen. The formation of - from B(OH) 3 would be expected therefore to occur via a similar initial reaction (attack by RO - or deprotonation of coordinated ROH) followed by a subsequent elimination of H 2O and the formation of a chelate coordination, and would therefore be related to the pKa of the alcohol. where the formation of hydroxide and/or other oxygen-containing species. 
The conversion of boric acid to borate (6.7.3) must occur through attack of hydroxide or the deprotonation of a coordinated water ligand, either of which is related to the pKa of water. The electron transfer is often affected by the presence of oxygen and/or surface. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
